 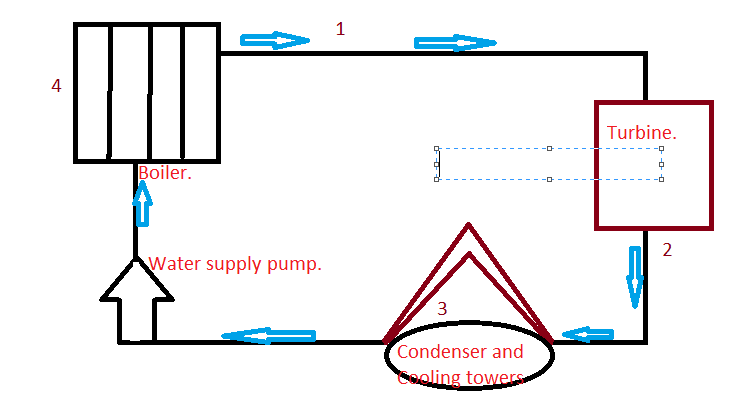 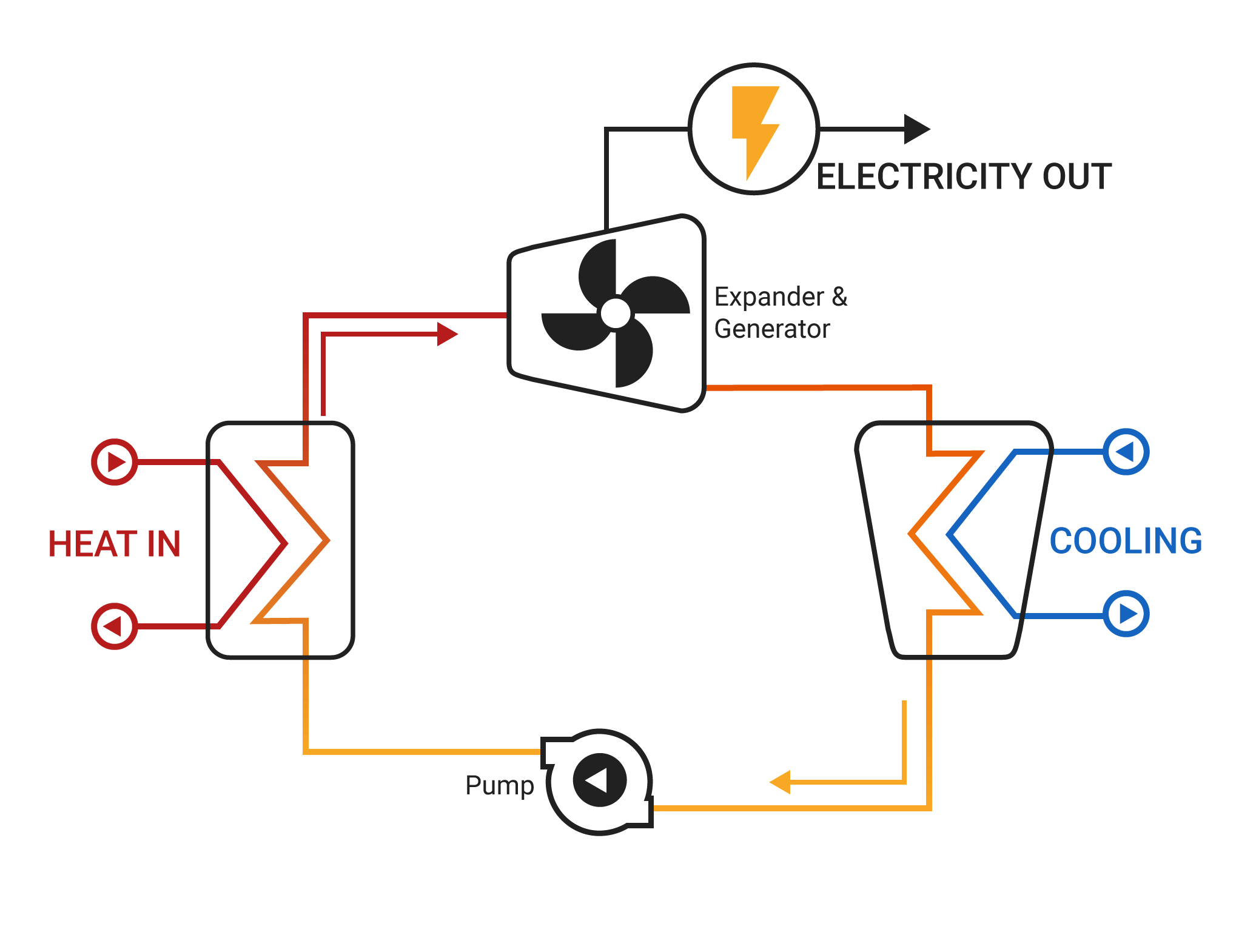
The main feature of the Rankine cycle, shown in Figure 31, is that it confines the isentropic compression process to the liquid phase only (Figure 31 points 1 to 2). Towards this end, the Rankine cycle was developed. It was desirable to construct a cycle that was as close to being reversible as possible and would better lend itself to the characteristics of steam and process control than the Carnot cycle did. Finally, a condenser designed to produce a two-phase mixture at the outlet (point1) would pose technical problems.Įarly thermodynamic developments were centered around improving the performance of contemporary steam engines. Second, this same is entropic compression will probably result in some pump cavitation in the feed system. First a great deal of pump work is required to compress a two phase mixture of water and steam from point 1 to the saturated liquid state at point 2. As shown, it has several problems which make it undesirable as a practical power cycle. Figure 30 shows a proposed Carnot steam cycle superimposed on a T-s diagram. Note that these numbers are less than 1/2 of the maximum thermal efficiency of the ideal Carnot cycle calculated earlier. Typical thermal efficiencies for fossil plants are on the order of 40% while nuclear plants have efficiencies of the order of 31%. As can be seen, the actual available energy (area under the 1-2-3-4 curve, Figure 29) is less than half of what is available from the ideal Carnot cycle (area under 1-2-4 curve, Figure 29) operating between the same two temperatures. Because of this, the average temperature at which we are adding heat is far below the maximum allowable material temperature. 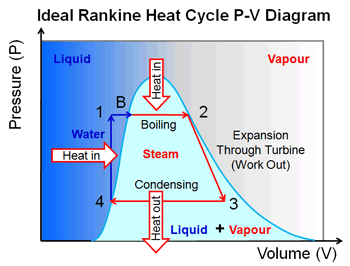
In reality, the nature of water and certain elements of the process controls require us to add heat in a constant pressure process instead (Figure 29, 1-2-3-4). If we wish to limit ourselves to operation at or below 2000 psia, it is readily apparent that constant heat addition at our maximum temperature of 1962R is not possible (Figure 29, 2 to 4). The working fluid is water, which places certain restrictions on the cycle. If the temperature of the heat sink is known, then thec hange in entropy does correspond to a measure of the heat rejected by the engineįigure 29 is a typical power cycle employed by a fossil fuel plant. From equation 1-37, one can see why the change in entropy can be defined as a measure of the energy unavailable to do work. Note that by developing materials capable of withstanding the stresses above 1962R, we could greatly add to the energy available for use by the plant cycle. From Figure 28 it can been seen that any cycle operating at a temperature of less than 1962R will be less efficient. Not all of this energy is available for use by the heat engine since a portion of it (qr) must be rejected to the environment.Īnd is equal to the area of the shaded region labeled available energy in Figure 28 between the emperatures 1962 and 520R. The energy added to a working fluid during the Carnot isothermal expansion is given by qs. We will do this by looking at the T-s diagrams of Carnot cycles using both real and ideal components. To understand why an efficiency of 73% is not possible we must analyze the Carnot cycle, thenc ompare the cycle using real and ideal components. Instead of being “wasted” by release into the ambient environment, sometimes waste heat (or cold) can be utilized by another process, or a portion of heat that would otherwise be wasted can be reused in the same process if make-up heat is added to the system (as with heat recovery ventilation in a building). Sources of waste heat include all manner of human activities, natural systems, and all organisms. Waste heat has lower utility (or in thermodynamics lexicon a lower exergy or higher entropy ) than the original energy source. The need for many systems to reject heat as a by-product of their operation is fundamental to the laws of thermodynamics. Waste heat or heat rejection is by necessity produced both by machines that do work and in other processes that use energy, for example in a refrigerator warming the room air or a combustion engine releasing heat into the environment. Thermodynamics Directory | Heat Transfer Directory Heat Rejection Waste Heat Thermodynamic Properties
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
